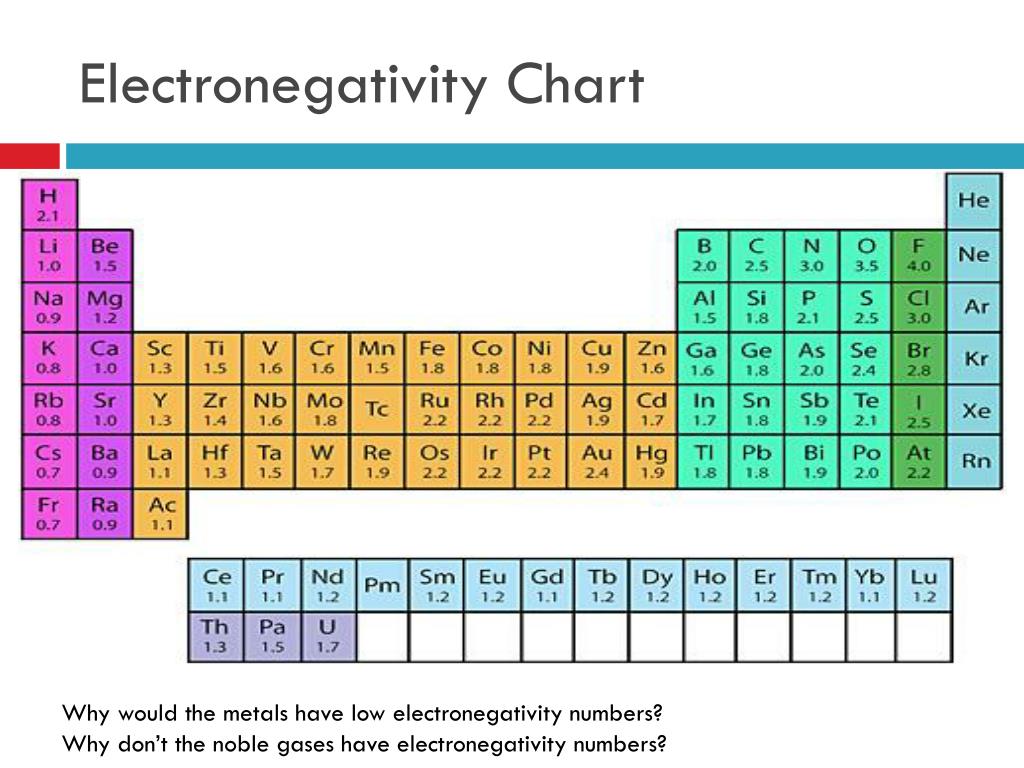
‘To our surprise, you cannot reconcile bond energies, both calculated and experimental, and the values of Pauling’s electronegativities,’ Oganov says. He asked his PhD student Tantardini to check whether their computational approach could produce atomic electronegativities at zero pressure. Oganov had been defining electronegativities of elements at high pressure to help explain exotic chemical phenomena taking place under these conditions. ‘Looking at Pauling electronegativities, you will see that in molybdenum borides or tungsten hydrides, metal atoms should be negatively charged, and non-metals positively charged, which is outrageous for chemical intuition,’ Oganov says. But Pauling’s definition sometimes doesn’t represent reality well. As such the idea often explains which chemical reactions the laws of thermodynamics will allow to work. It denotes how evenly or unevenly electrons are shared between atoms in a chemical bond. ‘Electronegativity is probably the most important chemical property of the elements,’ says Oganov. The new formula aligns better with whether reactions release or absorb energy, and avoids strange predictions that the original made for metal-containing modules. 
Artem Oganov and Christian Tantardini from Skolkovo Institute of Science and Technology, Moscow, made a simple-looking change to the equation originally devised by Linus Pauling in 1932. Researchers in Russia have modified the formula for the fundamental concept of electronegativity, making it more useful for predicting other aspects of chemistry. The electronegativities of the elements calculated using the formula created by Artem Oganov and Christian Tantardini Source: © Christian Tantardini and Artem R.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
